- Thyroid
- Different Molecular Phenotypes of Progression in BRAF- and RAS-Like Papillary Thyroid Carcinoma
-
Jinsun Lim, Han Sai Lee, Jiyun Park, Kyung-Soo Kim, Soo-Kyung Kim, Yong-Wook Cho, Young Shin Song
-
Endocrinol Metab. 2023;38(4):445-454. Published online July 18, 2023
-
DOI: https://doi.org/10.3803/EnM.2023.1702
-
-
 Abstract Abstract
 PDF PDF Supplementary Material Supplementary Material PubReader PubReader  ePub ePub
- Background
Papillary thyroid carcinoma (PTC) can be classified into two distinct molecular subtypes, BRAF-like (BL) and RASlike (RL). However, the molecular characteristics of each subtype according to clinicopathological factors have not yet been determined. We aimed to investigate the gene signatures and tumor microenvironment according to clinicopathological factors, and to identify the mechanism of progression in BL-PTCs and RL-PTCs.
Methods
We analyzed RNA sequencing data and corresponding clinicopathological information of 503 patients with PTC from The Cancer Genome Atlas database. We performed differentially expressed gene (DEG), Gene Ontology, and molecular pathway enrichment analyses according to clinicopathological factors in each molecular subtype. EcoTyper and CIBERSORTx were used to deconvolve the tumor cell types and their surrounding microenvironment.
Results
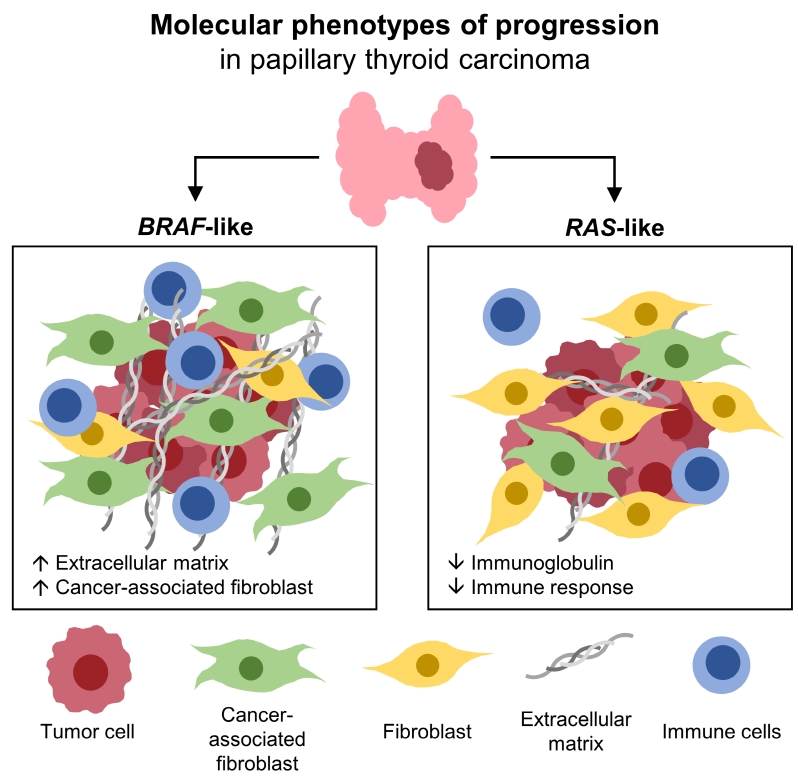
Even for the same clinicopathological factors, overlapping DEGs between the two molecular subtypes were uncommon, indicating that BL-PTCs and RL-PTCs have different progression mechanisms. Genes related to the extracellular matrix were commonly upregulated in BL-PTCs with aggressive clinicopathological factors, such as old age (≥55 years), presence of extrathyroidal extension, lymph node metastasis, advanced tumor-node-metastasis (TNM) stage, and high metastasis-age-completeness of resection- invasion-size (MACIS) scores (≥6). Furthermore, in the deconvolution analysis of tumor microenvironment, cancer-associated fibroblasts were significantly enriched. In contrast, in RL-PTCs, downregulation of immune response and immunoglobulin-related genes was significantly associated with aggressive characteristics, even after adjusting for thyroiditis status.
Conclusion
The molecular phenotypes of cancer progression differed between BL-PTC and RL-PTC. In particular, extracellular matrix and cancer-associated fibroblasts, which constitute the tumor microenvironment, would play an important role in the progression of BL-PTC that accounts for the majority of advanced PTCs.
- Adrenal Gland
- Aldosterone Immunoassay-Specific Cutoff Value for Seated Saline Suppression Test for Diagnosing Primary Aldosteronism
-
So Yoon Kwon, Jiyun Park, So Hee Park, So Hyun Cho, You-Bin Lee, Soo-Youn Lee, Jae Hyeon Kim
-
Endocrinol Metab. 2022;37(6):938-942. Published online December 6, 2022
-
DOI: https://doi.org/10.3803/EnM.2022.1535
-
-
1,807
View
-
194
Download
-
1
Web of Science
-
1
Crossref
-
 Abstract Abstract
 PDF PDF Supplementary Material Supplementary Material PubReader PubReader  ePub ePub
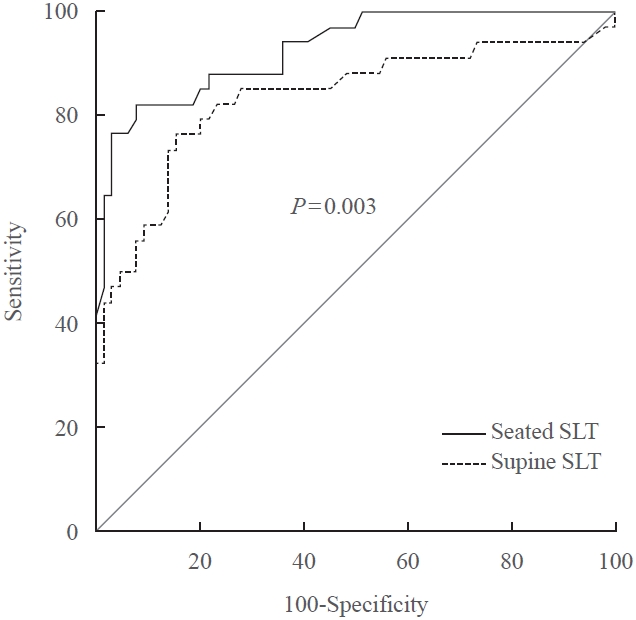
- A seated saline loading test (SLT) using liquid chromatography-tandem mass spectrometry (LC-MS/MS) is one of the most accepted confirmatory tests of primary aldosteronism. However, LC-MS/MS is time-consuming and is not widely available in diagnostic laboratories compared to immunoassay. With immunoassay, it is unknown whether SLT in the seated position is more accurate than that of the supine position, and a cutoff value of post-seated SLT plasma aldosterone concentration (PAC) must be established in the Korean population. Ninety-eight patients underwent SLT in both positions, and post-SLT PAC was measured by LC-MS/MS and radioimmunoassay. We confirmed primary aldosteronism if post-seated SLT PAC by LC-MS/MS exceeded 5.8 ng/dL. The area under the receiver operating characteristic curve was greater for seated than supine SLT (0.928 vs. 0.834, P=0.003). The optimal cutoff value of post-seated SLT by radioimmunoassay was 6.6 ng/dL (sensitivity 83.3%, specificity 92.2%).
-
Citations
Citations to this article as recorded by  - Investigating the cut-off values of captopril challenge test for primary aldosteronism using the novel chemiluminescent enzyme immunoassay method: a retrospective cohort study
Yuta Tezuka, Kei Omata, Yoshikiyo Ono, Kengo Kambara, Hiroki Kamada, Sota Oguro, Yuto Yamazaki, Celso E. Gomez-Sanchez, Akihiro Ito, Hironobu Sasano, Kei Takase, Tetsuhiro Tanaka, Hideki Katagiri, Fumitoshi Satoh
Hypertension Research.2024;[Epub] CrossRef
|